
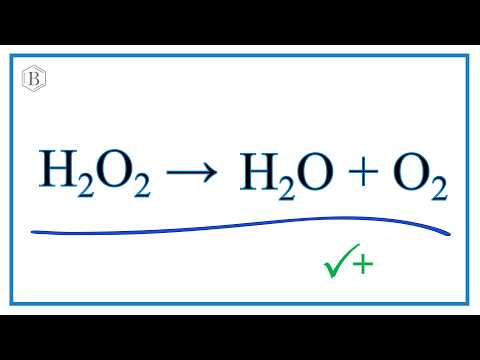
Comment équilibrer : H2O2 → O2 + H2O (peroxyde d'hydrogène, dioxygène, eau) | Physique-Chimie - YouTube

H2O+O2=H2O2 Balance the chemical equation. h2o+o2=h2o2 water and oxygen to form hydrogen peroxide - YouTube

Molécule Simple Du Peroxyde D'hydrogène H2O2 Illustration de Vecteur - Illustration du liquide, isolement: 40869920

Reduction of O2 to H2O and its free radical intermediates (A) Lewis... | Download Scientific Diagram

Hydrogen peroxide (H2O2) can act as oxidising as well as reducing agent in both acidic and alkaline media. From the following given reactions, select the option(s) in which reducing action of H2O2

Effect of (H2O)n (n = 1–3) clusters on H2O2 + HO → HO2 + H2O reaction in tropospheric conditions: competition between one-step and stepwise routes: Molecular Physics: Vol 117, No 4

SOLVED: Balance the following equation: H2O2 H2O + O2 H2O2 H2O + O2 2 H2O2 2 H2O + O2 2 H2O2 2 H2O + 3 O2 H2O2 2 H2O + O2


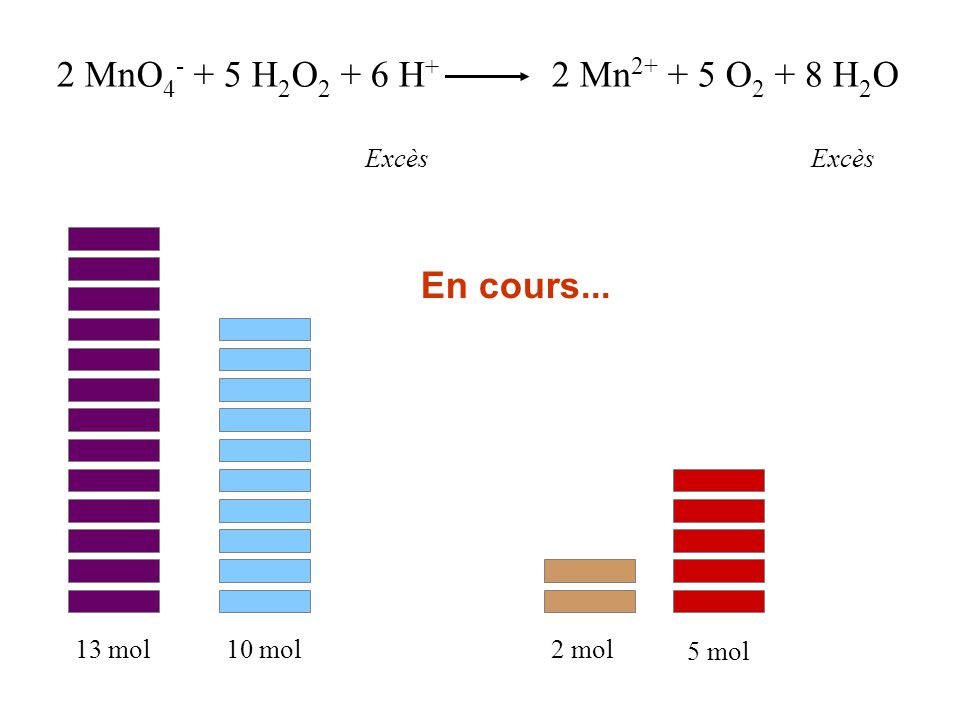










![SOLVED] I H2O2+O3→H2O+2O2II H2O2+Ag2O→2Ag+H2O+O2Role of hydroge - Self Study 365 SOLVED] I H2O2+O3→H2O+2O2II H2O2+Ag2O→2Ag+H2O+O2Role of hydroge - Self Study 365](https://static.tllms.com/ckeditor_assets/pictures/29060/content_1.jpg)
