![Write the reaction of action of heat on boric acid [H3BO3]? - Find 2 Answers & Solutions | LearnPick Resources Write the reaction of action of heat on boric acid [H3BO3]? - Find 2 Answers & Solutions | LearnPick Resources](https://www.learnpick.in/files/answerimages/5eb22de957ba09459f6fe1c23b3b4438.jpg)
Write the reaction of action of heat on boric acid [H3BO3]? - Find 2 Answers & Solutions | LearnPick Resources

Phase diagram of the H3BO3 + H2O system. Symbols represent experiemntal... | Download Scientific Diagram

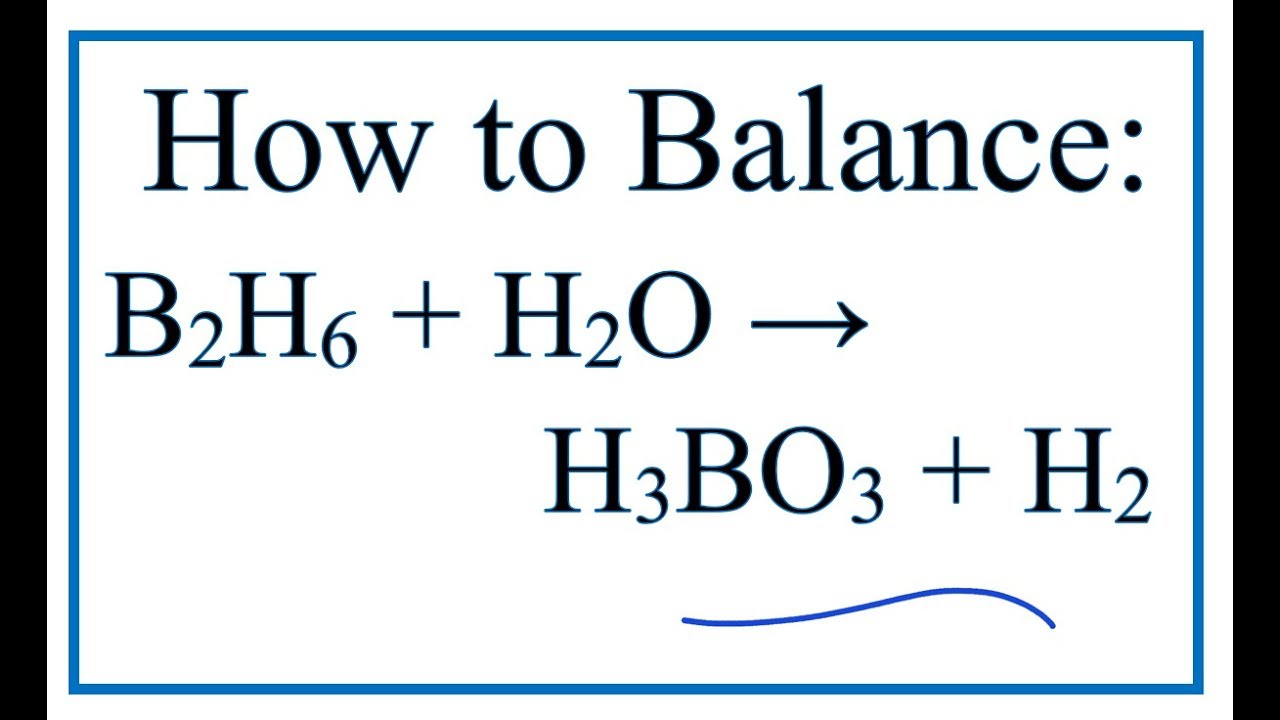
B2O3 + 3 H2O ---> 2 H3BO3 If diboron trioxide is reacted with water, the product is boric acid. What mass of boric acid is obtained from the full reaction. - ppt video online download

Differences of water activity, aw, and enthalpy of dilution, ΔdilHm, of... | Download Scientific Diagram

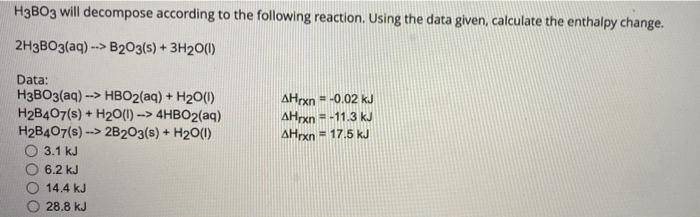
Calculate AH° the following reactions - 2H3BO3(aq) → B2O3(s) + 3H2O() Given that - (a) H3BO3(aq) → HBO2(aq) + H2O(), AHỤ = - 0.02 kJ (b) H2B4076) ▻ 2B0318) + H2O(, AH' =