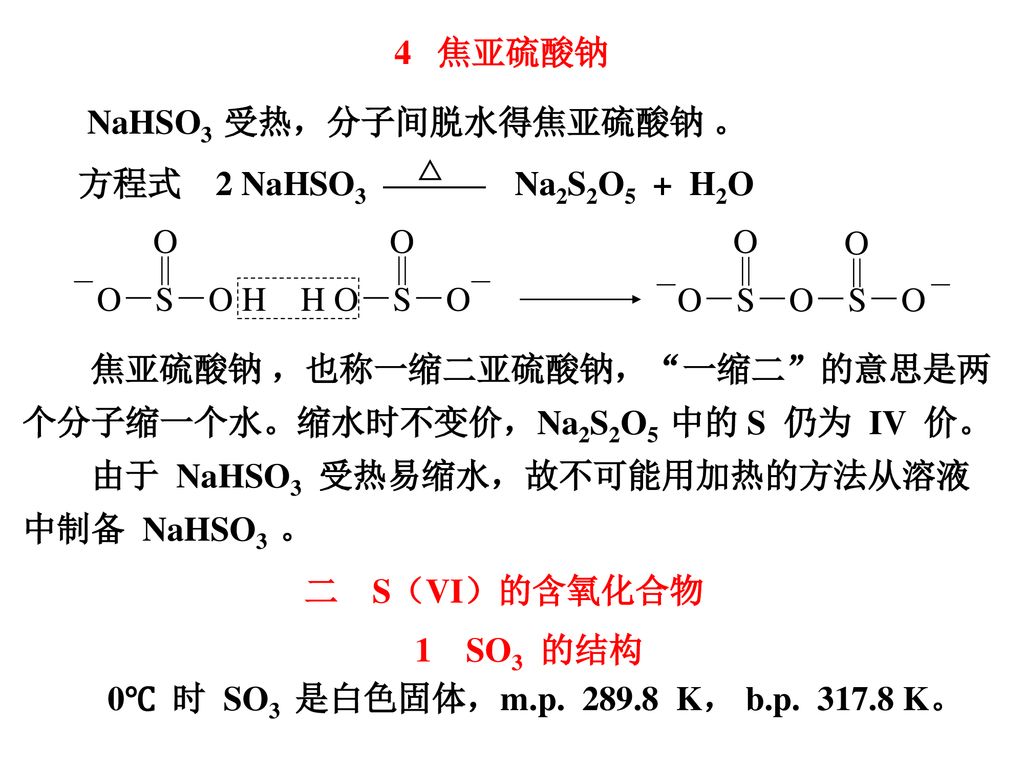
A solution of 12 in aqueous KI on being treated with an aqueous solution of Na2S2O3 gets decolourised. The reaction in question is: (a) Na2S2O3 + H2O +12 —> Lodometni stration Na2S204 +

SOLVED: Sulfur dioxide can be produced in the laboratory by the reaction of hydrochloric acid, HCl, and a sulfite salt such as sodium sulfite. The unbalanced reaction is shown below: HCl (g) +

Construction of β-Amino Sulfones from Sodium Metabisulfite via a Radical 1,4-Amino Migration | Organic Letters

SOLVED: i) What is the purpose of adding sodium metabisulfite to the organic solution containing benzyl alcohol? Demonstrate its function chemically: i) After above work out why sodium bicarbonate was added: Show