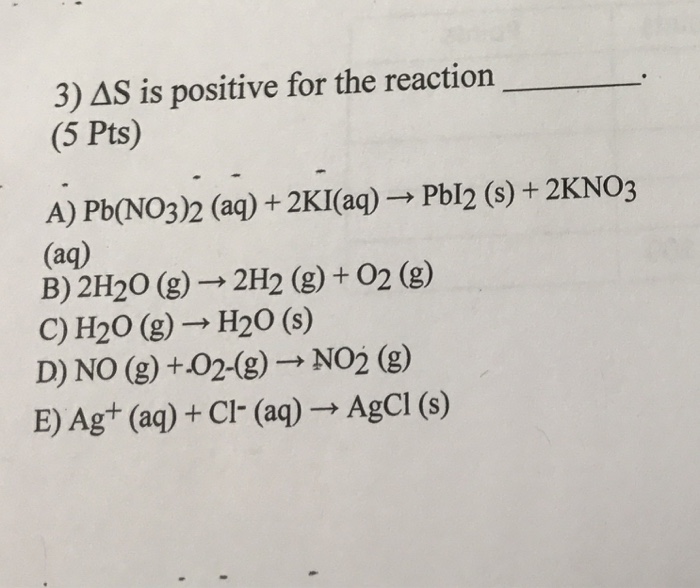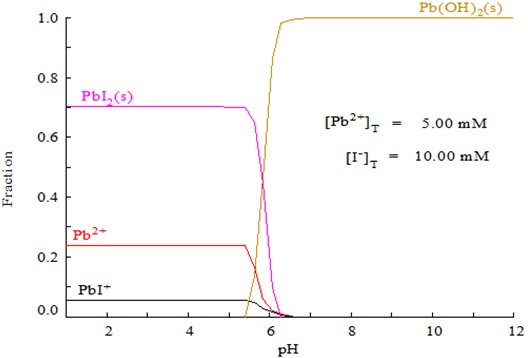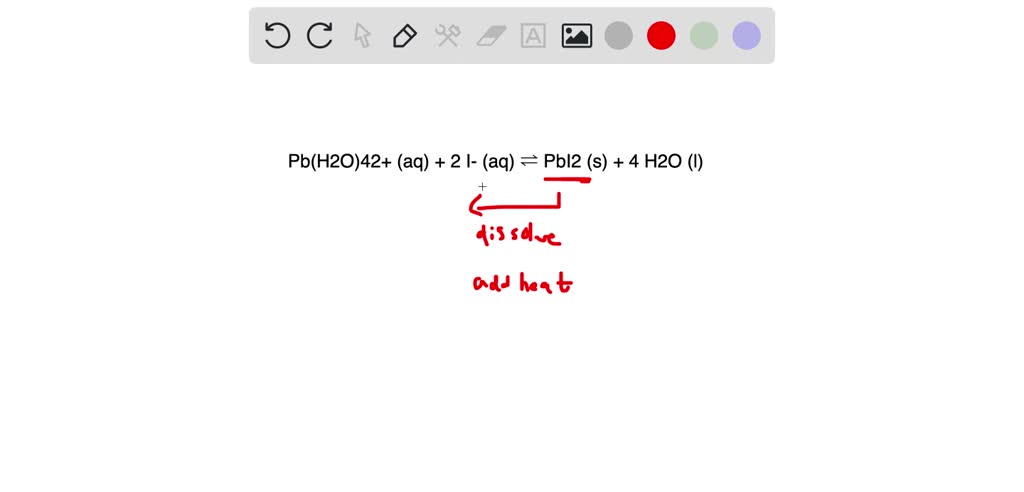
SOLVED: Pb(H2O)42+ (aq) + 2 I- (aq) ⇌ PbI2 (s) + 4 H2O (l) Say if the following statement about this equilibrium system is true or false:If the PbI2 (s) is slightly

Improvement of Colloidal Characteristics in a Precursor Solution by a PbI2-(DMSO)2 Complex for Efficient Nonstoichiometrically Prepared CsPbI2.8Br0.2 Perovskite Solar Cells | ACS Applied Materials & Interfaces

Scheme for the conversion from amorphous PbS to PbI2 using sublimated... | Download Scientific Diagram
![PDF] Facet-Dependent Control of PbI2 Colloids for over 20% Efficient Perovskite Solar Cells | Semantic Scholar PDF] Facet-Dependent Control of PbI2 Colloids for over 20% Efficient Perovskite Solar Cells | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/e86bab571167d5e9ddc42c29f2741c4cbbf07b6c/2-Figure1-1.png)
PDF] Facet-Dependent Control of PbI2 Colloids for over 20% Efficient Perovskite Solar Cells | Semantic Scholar

Controlling PbI2 Stoichiometry during Synthesis to Improve the Performance of Perovskite Photovoltaics | Chemistry of Materials
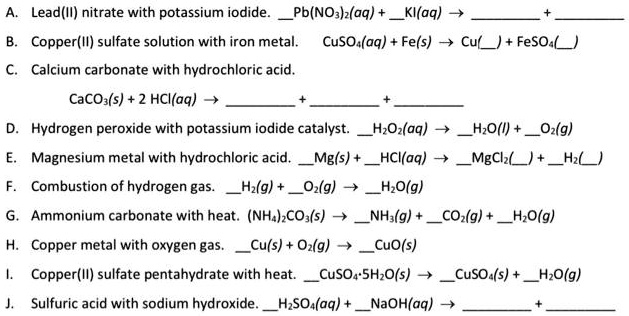
SOLVED: A. Lead(II) nitrate with potassium iodide. Pb(NO3)2(aq) + 2KI(aq) -> PbI2(s) + 2KNO3(aq) B. Copper(II) sulfate solution with iron metal. CuSO4(aq) + Fe(s) -> Cu(s) + FeSO4(aq) C. Calcium carbonate with

Few-Layer PbI2 Nanoparticle: A 2D Semiconductor with Lateral Quantum Confinement | The Journal of Physical Chemistry Letters

Enhancing efficiency and stability of perovskite solar cells through two-step deposition method with the addition of cesium halides to PbI2 precursor - ScienceDirect